Discussion on the Testing Method of Drug Stability Test Chamber
The Drug Stability Test Chamber (hereinafter referred to as the test chamber) provides a specific temperature and humidity environment for drug stability testing and accelerated testing. A large number of drug samples to be tested are stored inside the chamber. The temperature and humidity environment inside the chamber must meet the specified technical indicators before the test can be carried out. Its uniformity, fluctuation and other technical indicators are subject to authoritative testing by the quality and technical supervision department.


1 Working principle and technical indicators of the test chamber
The test chamber uses an electric heater as a heat source and is equipped with a refrigeration compressor to control the temperature. It uses an electric steam humidifier (using pure water or distilled water for water supply) and refrigeration and dehumidification equipment to control the humidity. Most Test Chambers can provide a temperature control range of 15℃~65℃ and a humidity control range of 20%RH~95%RH. For the stability test of drug preparations, the deviation between each point in the effective space of the drug stability test chamber and the set test temperature should be less than ±2℃. The humidity deviation should be less than ±5%RH.
2 Test method for temperature and humidity of the test chamber
Reference to "JJF 1101-2003 Environmental Test Equipment Temperature and Humidity Calibration Specifications" and combined with "2010GMP-Chinese Pharmacopoeia Stability Test Guidelines", the test method is formulated as follows:
2.1 Test items
The test items mainly involve the deviation, uniformity and fluctuation of temperature and humidity under the conditions of empty and full load of the test chamber. The following is a brief description of the test items.
Deviation: The difference between the average value of n measurements at the center of the test chamber and the average value of the temperature (relative humidity) displayed by the equipment.
Uniformity: The arithmetic mean of the difference between the highest temperature (relative humidity) and the lowest temperature (relative humidity) measured in n tests of the test chamber in a stable state.
Fluctuation: The change in the temperature (relative humidity) of the center of the chamber over time in a stable state, that is, half of the difference between the highest temperature (relative humidity) and the lowest temperature (relative humidity) at the center in n measurements, and is marked with a "±" sign.
2.2 Test duration and sampling cycle
The "JJF 1101-2003 Calibration Specification for Temperature and Humidity of Environmental Test Equipment" (hereinafter referred to as the calibration specification) stipulates that the test duration is 30 minutes, and the test data is recorded every 2 minutes. Considering that the stability test cycle of drug preparations in the test chamber is relatively long, with the shortest cycle being six months, a short 30 minutes is not enough to verify the stability index of the test chamber. The 24-hour test duration can better find some problems in the stability of the test chamber in the actual test work, which are not shown at all in the 30-minute test. We have also conducted a continuous test of up to 72 hours, and the test results are almost the same as the 24-hour test results. Therefore, we set the test duration to 24 hours for no-load and full-load (when the pharmaceutical company really needs it, it can be appropriately extended), and the recording interval is still 2 minutes mentioned in the calibration specification.
2.3 Selection of standard equipment for testing
The calibration specification requires that the temperature measurement standard should consist of a temperature sensor (usually a four-wire platinum thermal resistor) and a display instrument, with a time constant of less than 15s. The humidity measurement standard can be selected from digital ventilation psychrometer and barometer, digital hygrometer and dry and wet bulb thermometer. Considering that the test box is generally small in volume, some standard instruments are difficult to be tested in the box. For this reason, we selected a multi-channel high-precision temperature and humidity inspection instrument tested by the National Meteorological Metrology Station, with an expanded uncertainty of: temperature: 0.3℃ (k=2), humidity: 1.5%RH (k=2). The sensor used in this instrument is an integrated temperature and humidity design, which is small and light with high precision. Up to 14 sensors can collect data at the same time. The collector can sample at specified time intervals and store the sampling results to meet the needs of testing the test box.
2.4 Selection of the tested temperature and humidity points
The selection of temperature and humidity points is mainly based on user needs. The long-term stability test of general pharmaceutical preparations is 25℃, 60%RH; the accelerated test is 40℃, 75%RH.
2.5 Location and number of test points in the test chamber
The volume of the test chamber is small, generally less than 2m3. According to the calibration specification, when the equipment volume is less than 2m3, there are 9 temperature test points and 3 humidity test points. Considering that the test standard equipment we selected collects temperature and humidity at the same time, we choose 9 temperature and humidity test points here, distributed in the upper, middle and lower layers of the test chamber. The location of the test points is shown in Figure 1.
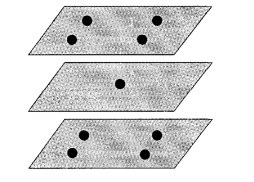
Figure 1
3 Data processing of test results
3.1 Temperature deviation calculation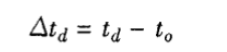
Where: △td—temperature deviation, °C;
t0 —average value of n measurements at the center point, °C;
td —average value of the temperature displayed on the test chamber, °C.
3.2 Temperature uniformity calculation
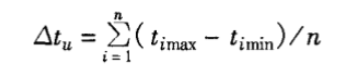
Where: △tu—temperature uniformity, ℃;
n—number of measurements;
timax—the highest temperature measured at each test point in the i-th time, ℃;
timin—the lowest temperature measured at each test point in the i-th time, ℃.
3.3 Calculation of temperature fluctuation
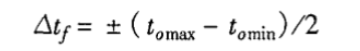
Where: △tf—temperature fluctuation, ℃;
tomax—highest temperature among n measurements of the center point, ℃;
tomin—lowest temperature among n measurements of the center point, ℃.
3.4 Calculation of relative humidity deviation
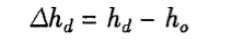
Where: △hd—humidity deviation, %RH;
ho—average value of n measurements at the center point, %RH;
hd —average value of humidity displayed on the test chamber, %RH.
3.5 Calculation of relative humidity uniformity
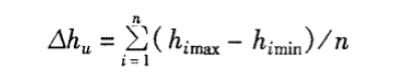
Where: △hu—humidity uniformity, %RH;
n—number of measurements;
himax—the highest humidity measured at each test point at the i-th time, %RH;
himin—the lowest humidity measured at each test point at the i-th time, %RH.
3.6 Calculation of relative humidity fluctuation
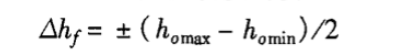
Where: △hf—humidity fluctuation, %RH;
homax—the highest humidity value among n measurements at the center point, %RH;
homin—the lowest humidity value among n measurements at the center point, %RH.
4 Issuance of test results
The test results should include the calculation results of deviation, fluctuation, and uniformity, and explain the standard instruments used for the test to prove that the test results are true and reliable. The calculation results of the test data are provided for reference by pharmaceutical companies to better carry out stability tests of drug preparations.
5 Conclusion
The test chamber has been tested and qualified by the manufacturer before leaving the factory, but with the long-term continuous operation of the test chamber, the performance of the temperature and humidity control equipment has declined, and some even cannot meet the environmental conditions required for the test. It is necessary to test and evaluate the overall performance of the chamber regularly. In addition, the test chamber is easier to reach the stability conditions of drug preparations when it is unloaded, but when it is fully loaded, it is affected by the load and the temperature and humidity control ability of the test chamber is significantly reduced, and even exceeds the temperature and humidity conditions required for the test. In this case, contact the manufacturer for repair in time, and perform the performance test of the test chamber again after the repair.













